The use of orthobiologic and regenerative therapies has increased dramatically over the past decade, yet misconceptions about these treatments and their target conditions remain. Although osteoarthritis (OA) is commonly conceptualized as an inflammatory disease, multiple research studies using inhibitors of cytokines such as TNF and IL-1 have failed to show effectiveness (Aitken, Laslett et al. 2018, Kloppenburg, Ramonda et al. 2018).
Reframing OA as a chronic wound with a stalled healing immune response explains the failures of these prior cytokine studies. The osteoarthritic joint remains locked in a low-grade state of inflammation, with the release of degradative enzymes such as matrix metalloproteinases (MMPs). This non-healing wound leads to collagen breakdown, hyaluronic acid fragmentation, and persistent inflammation through a feed-forward cycle. To effectively treat chronic conditions like OA a paradigm shift from fighting inflammation to resolving it through immune stimulation is necessary. If successful healing depends on actively resolving inflammation, could orthobiologic therapies be the tools capable of triggering this process?
In Healing Joints and Nerves, this emerging paradigm shift in the treatment of OA and other chronic pain conditions is brought to life and made accessible to a broader audience.
Platelet-rich plasma
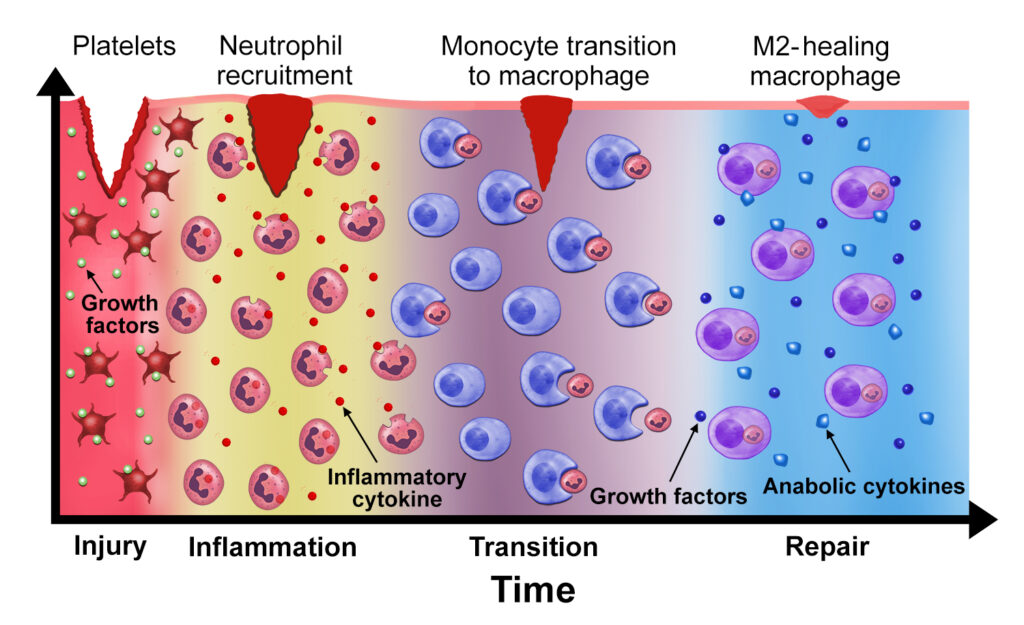
Therapeutic immune stimulation can be harnessed through orthobiologic therapies such as platelet-rich plasma (PRP) and mesenchymal stem cells (MSCs). PRP contains platelets, their growth factors, and variable concentrations of leukocytes. When the platelet dose is sufficient, this combination of factors is capable of reactivating a stalled healing cascade and reducing the pain associated with OA (Figure 1). At its core, PRP isn’t an anti-inflammatory treatment. It uses acute inflammation to act as an immune stimulant. The critical role of immune activation is further supported by recent studies demonstrating that PRP is likely to be effective only when the platelet and leukocyte doses are robust (Bansal, Leon et al. 2021, Berrigan, Bailowitz et al. 2025). Variability and lack of processing standards continue to be problematic for PRP use in clinical practice.
Mesenchymal “stem cells”
Likewise, MSCs also work through immune modulation. MSCs, originally isolated from bone marrow in the 1980s by Dr. Arnold Caplan, demonstrate the ability to differentiate into chondrocytes in cell culture. Early laboratory experiments generated great enthusiasm for the potential of cartilage regrowth, based on these in vitro studies. However, the role of MSCs in vivo differs significantly from their activity in a lab culture dish. Once injected, MSC viability lasts from 24 hours to several weeks, depending on the injection route and the rate of macrophage phagocytosis (de Witte, Luk et al. 2018, Satué, Schüler et al. 2019). Contrary to Dr. Caplan’s initial hypothesis, the pain relief from MSC injections doesn’t come from cartilage regrowth. Instead, pain relief results from MSCs’ secreted factors (Chen, Park et al. 2015, de Witte, Luk et al. 2018) and from macrophage activation (Guo, Imai et al. 2017).
Dr. Caplan increasingly understood the immune-based mechanisms of MSCs, and he eventually authored the 2017 editorial, “Mesenchymal Stem Cells: Time to Change the Name!” (Caplan 2017). He argued we should call these cells “medicinal signaling cells” to better reflect their biological functions. Despite his pleading, MSCs are still commonly referred to as “stem cells” in the lay press and medical publications. Although MSCs have shown analgesic benefits in several clinical studies, a recent multi-center, randomized clinical trial has called into question their potential advantages over traditional treatments such as corticosteroid injection (Mautner, Gottschalk et al. 2023).
Autologous Conditioned Serum (ACS)
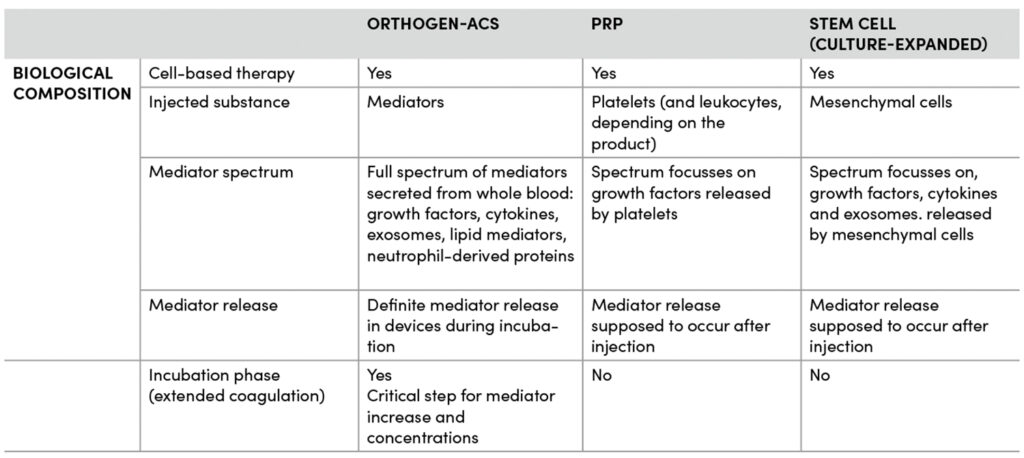
Immune-based methods to resolve inflammation are also a core element of the orthobiologic therapy, autologous conditioned serum (ACS). ACS represents the physiological whole-blood secretome, encompassing the full spectrum of mediators released by all blood cells during an incubation phase- a process that mirrors the crucial phase of natural tissue healing. This generates a biologically potent composition including growth factors, cytokines, lipid mediators, neutrophil-derived factors and exosomes which leads to effective tissue regeneration and pain resolution. Therapy concepts including this particular ACS are invented by a German biotech company called Orthogen. The extended benefits are shown in over 45 clinical studies of ACS (Baltzer, Moser et al. 2009, Baltzer, Ostapczuk et al. 2013, Damjanov, Barac et al. 2018, Hang N 2025) including indications for OA, spinal and nerve tissue diseases and other pathologies. Because benefits of Orthogen-ACS appeared to extend beyond the duration of growth factors and anti-inflammatory cytokines such as IL-1, Dr. Buchheit and research teams pursued additional mechanistic studies. Using laboratory models of neuropathy and chronic pain, the scientific teams demonstrated that ACS resolved inflammation, provided lasting pain relief, and even improved nerve function (Buchheit, Huh et al. 2023). The mediator profile of ACS is well characterized and recent analyses have demonstrated a marked increase in exosomes. When tested for function, the exosome contribution to ACS was significant, supporting long-term pain relief and tissue homeostasis through targeted intercellular signaling.
The unique and highly activated secretome profile including the diverse combination of factors in ACS has the ability to reduce the catabolic effects of corticosteroids (CS). Concerns about the potential negative impact of repeated CS injections have increased in recent years (McAlindon, LaValley et al. 2017). However, the side effects of CS appear to be markedly reduced with the addition of ACS. Combined treatment appears to enhance pain relief and reduce the risk of tissue injury associated with corticosteroids (Damjanov, Barac et al. 2018). By addressing acute pain instantly while simultaneously stimulating the body’s natural regenerative processes, it offers both short-term relief and long-term healing that can promote lasting effects.
There is a clear paradigm shift in treating OA and other chronic pain conditions. OA isn’t simply “wear and tear,” nor is it primarily an inflammatory condition like autoimmune diseases. Instead, OA is a chronic wound that requires treatment through immune stimulation and activation of the healing process. Regenerative therapies such as PRP, MSCs, and ACS do not work by “fighting” inflammation. They stimulate innate, immune-based repair mechanisms through various pathways. PRP delivers growth factors. MSCs promote macrophage activation. ACS functions through high concentrations of growth factors, lipid mediators, inflammation-resolving cytokines, and exosomes. Collectively, these mechanisms restore joint balance, encourage tissue repair, and relieve pain.
Relevant Recent Publications:
Buchheit T, Huh Y, Maixner W, Cheng J,
Ji RR. Neuroimmune modulation of pain and regenerative pain medicine. J Clin Invest. 2020;130(5):2164-76. PubMed PMID: 32250346
Buchheit T, Hunt C, Eldrige J, Eshraghi Y, Souza D. Product characteristics should be reported in all biological therapy publications. Reg Anesth Pain Med. 2022. PubMed PMID: 35318261
Buchheit T, Huh Y, Breglio A, Bang S, Xu J, Matsuoka Y, et al. Intrathecal administration of conditioned serum from different species resolves Chemotherapy-Induced neuropathic pain in mice via secretory exosomes. Brain Behav Immun. 2023. PMID: 37150265
Buchheit T, Hunt C, Eldrige J, Eshraghi Y, Souza D. Autologous Conditioned Plasma is not Platelet-Rich Plasma. Reg Anesth Pain Med. 2026. PMID: 41672585
Buchheit, T: Healing Joints and Nerves: Immune Stimulation and the New Science of Regenerative Therapies. Bull Publishing Company. 2026
References
Aitken, D., L. L. Laslett, F. Pan, I. K. Haugen, P. Otahal, N. Bellamy, P. Bird and G. Jones (2018). „A randomised double-blind placebo-controlled crossover trial of HUMira (adalimumab) for erosive hand OsteoaRthritis – the HUMOR trial.“ Osteoarthritis Cartilage 26(7): 880–887.
Baltzer, A. W. A., C. Moser, S. A. Jansen and R. Krauspe (2009). „Autologous conditioned serum (Orthokine) is an effective treatment for knee osteoarthritis.“ Osteoarthritis and Cartilage 17(2): 152–160–160.
Baltzer, A. W. A., M. S. Ostapczuk, D. Stosch, F. Seidel and M. Granrath (2013). „A New Treatment for Hip Osteoarthritis: Clinical Evidence for the Efficacy of Autologous Conditioned Serum.“ Orthopedic Reviews 5(2): e13.
Bansal, H., J. Leon, J. L. Pont, D. A. Wilson, A. Bansal, D. Agarwal and I. Preoteasa (2021). „Platelet-rich plasma (PRP) in osteoarthritis (OA) knee: Correct dose critical for long term clinical efficacy.“ Sci Rep 11(1): 3971.
Berrigan, W. A., Z. Bailowitz, A. Park, A. Reddy, R. Liu and D. Lansdown (2025). „A Greater Platelet Dose May Yield Better Clinical Outcomes for Platelet-Rich Plasma in the Treatment of Knee Osteoarthritis: A Systematic Review.“ Arthroscopy 41(3): 809–817.e802.
Buchheit, T., Y. Huh, A. Breglio, S. Bang, J. Xu, Y. Matsuoka, R. Guo, A. Bortsov, J. Reinecke, P. Wehling, T. Jun Huang and R. R. Ji (2023). „Intrathecal administration of conditioned serum from different species resolves Chemotherapy-Induced neuropathic pain in mice via secretory exosomes.“ Brain Behav Immun 111: 298–311.
Caplan, A. I. (2017). „Mesenchymal Stem Cells: Time to Change the Name!“ STEM CELLS Translational Medicine 6(6): 1445–1451–1451.
Chen, G., C. K. Park, R. G. Xie and R. R. Ji (2015). „Intrathecal bone marrow stromal cells inhibit neuropathic pain via TGF-beta secretion.“ J Clin Invest 125(8): 3226–3240.
Damjanov, N., B. Barac, J. Colic, V. Stevanovic, A. Zekovic and G. Tulic (2018). „The efficacy and safety of autologous conditioned serum (ACS) injections compared with betamethasone and placebo injections in the treatment of chronic shoulder joint pain due to supraspinatus tendinopathy: a prospective, randomized, double-blind, controlled study.“ Med Ultrason 20(3): 335–341.
de Witte, S. F. H., F. Luk, J. M. Sierra Parraga, M. Gargesha, A. Merino, S. S. Korevaar, A. S. Shankar, L. O’Flynn, S. J. Elliman, D. Roy, M. G. H. Betjes, P. N. Newsome, C. C. Baan and M. J. Hoogduijn (2018). „Immunomodulation By Therapeutic Mesenchymal Stromal Cells (MSC) Is Triggered Through Phagocytosis of MSC By Monocytic Cells.“ Stem Cells 36(4): 602–615.
Guo, W., S. Imai, J. L. Yang, S. Zou, M. Watanabe, Y. X. Chu, Z. Mohammad, H. Xu, K. D. Moudgil, F. Wei, R. Dubner and K. Ren (2017). „In vivo immune interactions of multipotent stromal cells underlie their long-lasting pain-relieving effect.“ Sci Rep 7(1): 10107.
Hang N, H. G., Wehling J, Reinecke J, Hang M (2025). „Orthogen Autologous Conditioned Serum: An Update of the currently published Clinical Studies.“ Medical research Archives 13.
Kloppenburg, M., R. Ramonda, K. Bobacz, W. Y. Kwok, D. Elewaut, T. W. J. Huizinga, F. P. B. Kroon, L. Punzi, J. S. Smolen, B. Vander Cruyssen, R. Wolterbeek, G. Verbruggen and R. Wittoek (2018). „Etanercept in patients with inflammatory hand osteoarthritis (EHOA): a multicentre, randomised, double-blind, placebo-controlled trial.“ Ann Rheum Dis 77(12): 1757–1764.
Mautner, K., M. Gottschalk, S. D. Boden, A. Akard, W. C. Bae, L. Black, B. Boggess, P. Chatterjee, C. B. Chung, K. A. Easley, G. Gibson, J. Hackel, K. Jensen, L. Kippner, C. Kurtenbach, J. Kurtzberg, R. A. Mason, B. Noonan, K. Roy, V. Valentine, C. Yeago and H. Drissi (2023). „Cell-based versus corticosteroid injections for knee pain in osteoarthritis: a randomized phase 3 trial.“ Nat Med 29(12): 3120–3126.
McAlindon, T. E., M. P. LaValley, W. F. Harvey, L. L. Price, J. B. Driban, M. Zhang and R. J. Ward (2017). „Effect of Intra-articular Triamcinolone vs Saline on Knee Cartilage Volume and Pain in Patients With Knee Osteoarthritis: A Randomized Clinical Trial.“ JAMA : the journal of the American Medical Association 317(19): 1967–1975.
Satué, M., C. Schüler, N. Ginner and R. G. Erben (2019). „Intra-articularly injected mesenchymal stem cells promote cartilage regeneration, but do not permanently engraft in distant organs.“ Sci Rep 9(1): 10153.
Buchempfehlungen
Healing Joints and Nerves (HJN)
Autor: Dr. Thomas Buchheit
ISBN: 978-1945188558
Autoren
,PhD, ist außerordentlicher Professor in der Abteilung für Anästhesiologie in Duke, North Carolina, USA und Direktor des Duke Regenerative Pain Therapies Program.