The goal of regenerative medicine is to rebuild diseased or damaged body tissue, with the aim of creating tissue with biological properties that are comparable to healthy tissue. This is in contrast to the traditional approach of repair, which seeks to restore normal function to diseased or damaged tissue but without restoring the tissue itself in terms of quality and quantity as in the Latin restitutio ad integrum (restoration to the original condition). This often ultimately results in failure and in artificial joint replacement, for example. Regenerative medicine seeks to achieve improved outcomes using biotechnological techniques, biological methods and stimulation and modulation of natural healing and to restore tissue homeostasis and adaptation and self-healing mechanisms.
Orthopaedics and regenerative medicine
The translation of preclinical results in regenerative research into products that can be used in clinical practice is slow. The reasons range from authorisation issues to ethical discussions about gaps in scientific understanding through to psychosocial issues. Nonetheless, orthopaedics can be viewed as a pioneer in the clinical application of regenerative medical devices. Orthopaedics and traumatology is a broad field of regenerative medicine with the restoration of musculoskeletal structures, such as the menisci, cartilage, bone and intervertebral discs. Given the high prevalence of injury in athletes, regenerative medicine is playing an increasingly important role in sports orthopaedics. This includes, on the one hand, tissue engineering, which involves the use of cultured cells of various origins on substrates and the addition of growth factors to support tissue regeneration and, on the other, the use of both complex blood derivatives (e.g. platelet and cell concentrates with a high proportion of mesenchymal stem cells (MSCs) from bone marrow or adipose tissue) and individual factors such as vesicles or small molecules that intervene in healing and regeneration cascades. The methods have a low adverse effect profile and are often better tolerated than traditional pain and inflammation inhibitors, which furthermore provide only symptomatic relief and do not support any regenerative potential. In sports medicine in particular, the natural regeneration potential of the mostly young patients is high and should be researched more intensively.
General strategies in regenerative medicine
In general, regenerative strategies are based on 4 cornerstones:
- cells
- a supporting matrix (biomaterial)
-
signals for tissue and cell differentiation
-
and environmental factors, such as biomechanical stimuli.
Cells and their source
Cells are the first cornerstone for regenerative medicine applications. Autologous cell transplants, as in chondrocyte transplantation, are already being used. The evidence for ACT has markedly improved and there are randomised studies confirming by biopsy and MRI the clinical efficacy of the method with restoration of the cartilaginous joint surface in isolated cartilage defects. Long-term studies with a follow-up period of up to 20 years have also confirmed the sustained effect of ACT. In approx. 75 % of cases, the morphology of the joint cartilage is substantially regenerated, which also supports joint longevity and, compared to microfracture, results in improved outcomes, especially beyond the five-year threshold. Just the microfracture-induced bleeding alone results in repair tissue consisting of mixed fibrous tissue and, as recent studies have shown, often in increasing bone formation in the defect, which thins out the cartilage above and ultimately results in failure.
The above impressively demonstrates that repair methods such as microfracture are clinically successful in the short term but are unable to treat cartilage defects in the long term. It is essential, therefore, that cell transplantation be used for large defects in particular, not least because microfracture also negatively affects the outcome of any subsequent cartilage surgery in the long term. Despite these successes with ACT, the logistics, administration and technology costs are so high that their cost-effectiveness is hard to justify. These criticisms, however, always have to be seen against the background of joint longevity and the associated improved quality of life and social health economics. Modern approaches are increasingly focusing on stem cells and progenitor cells. Stem cells from bone marrow, umbilical cord blood and adipose tissue have long been used in clinical practice; embryonic stems cells would be another very promising option but for the ethical issues involved. A modern alternative to embryonic stem cells might be induced pluripotent stem cells (iPSC). These are primarily somatic cells that have been reprogrammed to an embryonic stem cell-like state.
Matrices
Matrices are the second cornerstone of regenerative medicine; they are primarily used to provide a support and attach the inserted cells at the defect site. They are thus a scaffold on which new tissue can form. Modern matrices actively emit signals to promote the regeneration process and accordingly provide impulses for regeneration, which can be biological, chemical, or physical. How they emit these signals depends on their design. Modern, smart matrices react to environmental stimuli, whereas more traditional matrices, which are typically absorbable, release their factors as they are absorbed. Matrices consist of a wide variety of natural and synthetic materials and are often copolymers of different components. Natural materials are natural collagens, hyaluronates and fibrin, which are sometimes manufactured recombinantly. Synthetic biomaterials are polylactides and polycaprolactones or combinations of the two. The type of material selected depends on a wide variety of factors, such as porosity, biocompatibility and absorption rate. Modern matrices may also have a micro or nano structure or even be manufactured according to the individual anatomy. In this instance, bioprinting allows cells and growth factors to be integrated into the individually manufactured construct. To support the minimally invasive character of modern regenerative approaches overall, increasing use is also being made of matrices made of injectable, self-hardening gels or pastes.
Signals for differentiation
Signals/morphogenetic stimuli that stimulate cells to differentiate into specific tissue are the third cornerstone for regenerative medicine applications. The best known example of this are growth factors such as transforming growth factor beta (TGF-β), which plays an essential role in chondrogenesis. Alongside other stimuli such as transcription factors, trophic factors and small active molecules, the environmental milieu (e.g. hypoxia) can also initiate differentiation processes. A common problem here is that these factors can hardly ever be applied systemically but usually have to be applied locally at the site to be regenerated for days and even weeks. Local application over a prolonged period is the aim of smart scaffolds that are programmed to release such factors. However, to date it is often unclear what factor is required precisely when, in what concentrations it should be applied and what precise form the kinetics should take.
One pragmatic approach which avoids this problem altogether involves once more MSCs and other substances harvested from blood such as platelet-rich plasma (PRP). The abundance of individually different trophic factors automatically associated with these products saves the user the task of providing the precise concentration/isolating the individual factors. But this also turns a critical eye on blood products such as ACP and PRP. Their clinical efficacy has been demonstrated in both destroyed tissue and degenerative processes such as osteoarthritis. The mixture of anti-inflammatory, immunomodulatory and regenerative factors can act on the healing cascade and support tissue healing. With chronic pathologies in particular, the introduction of blood components reactivates healing processes through the release of platelet factors and with mesenchymal stem cell-derived microvesicles and can make good a failed attempt at healing that has become chronic.
Mechanical stimuli
Mechanical stimuli are the fourth cornerstone for the success of regenerative methods. They are crucial for the function and development of skeletal structures. The mechanical influence plays an essential role particularly in the initial differentiation phase but also in the later remodelling phase of healing. The use of continuous passive motion devices (CPM) plays an important role in the rehabilitation of cartilage defects. Much movement, little loading is the motto here and ensures optimum cartilage formation post cell transplantation. Another interesting application of mechanical stimuli to promote bone regeneration is extracorporeal shock wave therapy (ESWT). This is essentially also a regenerative measure to mechanically reactivate stalled healing processes using shock waves. The field of mechanical stimuli would seem to be a fertile ground for innovative rehabilitation protocols and might come more to the fore in regenerative medicine due to the above mechanisms.
——————————————————————————
SPECIFICS: Blood-derived products in regenerative orthopaedic sports medicine
The physiological support of blood-derived products for tissue repair and regeneration is attracting attention for many applications in regenerative medicine. Platelet-rich plasma (PRP) is one of the best known blood products commonly used as a supplement in in-vitro cell cultures and for therapeutic applications. The quantity of growth factors and cytokines available in the platelet alpha granules in PRP provides all the necessary anabolic factors to maintain proliferation, differentiation and cell phenotypes. Deviations that arise due to different PRP manufacturing protocols are responsible for the large number of different platelet-rich plasma products in laboratories globally.

Scientific value of PRP etc in tendon injuries
Tendon disorders of any kind make high demands in terms of diagnostics, treatment, rehabilitation and prevention at a fundamental level and often require interdisciplinary collaboration. Interest in and the use of orthobiologics (e.g. platelet-rich plasma and autologous conditioned plasma – PRP and ACP) has steadily grown in the last decade. Orthobiologics and the procedures followed for their application are often mentioned in the context of tendon injuries. The results of a current survey in the AOSSM (American Orthopaedic Society for Sports Medicine) confirm just how frequently they are used in routine clinical practice. Tendon injuries, together with osteoarthritis, are the second most common indication for the use of platelet-rich plasma. However, in this context it is viewed critically that with its increasing popularity the indications for its use are not always based on scientific evidence and therapeutic efficacy.
Acute tendon injuries and those due to overuse, together with functionally related structures (bone insertion areas, surrounding synovial tissue, myofascial interfaces), are one of the most common injuries and clinical pictures in athletes across all sports and ages. Epidemiologically, the (weight-bearing) tendons of the lower extremities (e.g. the Achilles tendon, patellar tendon) in particular are commonly involved. A large number of injuries in sports can be ascribed to different forms of overuse with or without concomitant risk factors. Fundamental knowledge of the pathogenesis is essential to understand damage mechanisms and be able to address them adequately and treat them successfully and appropriately depending on the stage. Where the aetiopathogenesis cannot be fully explained, an interaction between a change in metabolic activity (including tenocyte activity), a change in the structural integrity of the tendon and the presence of more or less inflammatory metabolic disorders can be assumed. The individually selected treatment methods share the goal of clinically identifying and improving impaired tendon function in a continuous and progressive treatment process. Careful selection and structuring of the treatment methods is essential here.
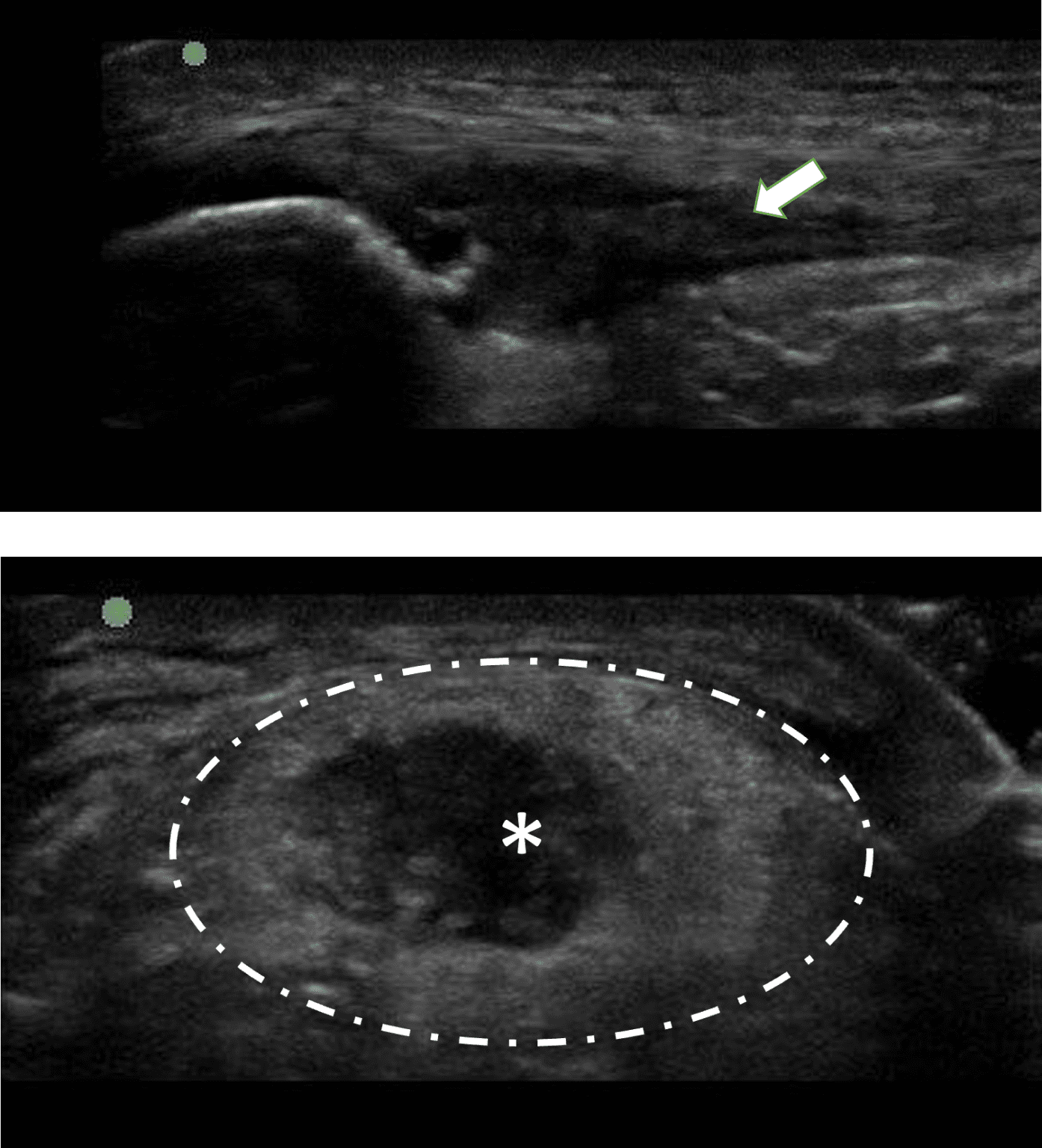
Application and/or infiltration as a procedure routinely available in clinical practice
When PRP etc were first used, the available evidence was inadequate due to the inadequate methodological quality of the studies then available. The at times method-related limitations of these studies and the different methods used to collect PRP resulted in different compositions and dosages. Moreover, the application and follow-up protocols were not uniform and still hinder scientific analysis and thus ultimately the assessment of this treatment method today. In principle, the cytokines and growth factors found in PRP can enhance the inflammatory and healing process. PRP, applied at the correct time and in the correct work-up, might thus have a positive effect on tissue regeneration. However, if the timing and work-up are inadequate, then the opposite effect may be triggered. It has now been generally established that the clinical efficacy of PRP depends on both the biological milieu (application as a supplement perioperatively as opposed to purely conservatively) and localisation of the application. The question therefore is no longer whether PRP is generally useful (e.g. irrespective of localisation and severity) but whether it should be applied in each individual case.
Positive effects for the patellar tendon, rotator cuff and radiohumeral epicondylopathy
The scientific data currently available demonstrate positive therapeutic effects, particularly for the patellar tendon, rotator cuff and radiohumeral epicondylopathy and for perioperative application in Achilles tendon reconstruction. Application of PRP for Achilles tendinopathy does not appear to be superior to other treatment methods and, based on current knowledge, cannot be fully recommended.
Summary of the use of orthobiologics for tendinopathy
- Heterogeneous data available on clinical efficacy – biological milieu and localisation are crucial
-
Indication must take into account the localisation and be adjusted to the underlying pathology
-
Solid data available: patellar tendon, rotator cuff, radiohumeral epicondylopathy, Achilles tendon reconstruction
-
No monotherapy, no unique selling point!
NB: The indication for infiltration treatment in tendinopathy should always be subject to strict scrutiny irrespective of the active substance applied. The user is in each case responsible for the infiltration and must always check what specific treatment goal is being pursued with the infiltration. In tendinopathy, the purpose of infiltrations (of any kind) is not to maintain sports activity or weight-bearing capacity at a symptomatic level. The goal of all those involved in the treatment should always be to enable, within a continuous rehabilitation process, a safe return to training and competition in line with the severity of the damage.
First published: GOTS* newsletter
GOTS*: The trinational (Germany, Austria, Switzerland) Society for Orthopaedic and Trauma Sports Medicine (GOTS) is the largest European association of sports orthopaedic and sports trauma specialists. It is the first point of contact in the care of sports injuries and a guarantee of quality in sports trauma care. Its goal is to improve the understanding of sports loads and injuries in order to maintain musculoskeletal function and quality of life. To this end, GOTS promotes training and continuing education, research and the sharing of information and expertise internationally among doctors active in sports orthopaedics and sports trauma and professional groups in related specialisms.
Autoren
ist Verbandsarzt der Deutschen Triathlon Union (DTU). Neben der klinischen Tätigkeit im Zentrum für Muskuloskelettale Chirurgie (OZMC) des Klinikums Osnabrück beschäftigt er sich wissenschaftlich schwerpunktmäßig mit Muskel- und Sehnenverletzungen und kann auf diesem Gebiet eine Vielzahl internationaler Publikationen vorweisen. Er ist Vorstandsmitglied der GOTS, Mitglied des GOTS-Komitees Muskel, Mitglied im „Muscle Research Center Erlangen“ (MURCE) und wiss. Beirat der sportärztezeitung.
ist Facharzt für Orthopädie und orthopädische Chirurgie. Er leitet das Zentrum für Regenerative Medizin und das Department für Gesundheitswissenschaften, Medizin, Forschung an der Donau Universität Krems, samt Professur für Tissue Engineering. Daneben ist er am Uni-Klinikum Krems an der orthopädischen Abteilung, mit Schwerpunkt Sportorthopädie und Knorpelchirurgie tätig. Seit 1992 in der GOTS, war er u.a. bereits deren Präsident und Vizepräsident Österreichs und war 2025 Kongresspräsident des GOTS-Kongresses in Krems sowie Präsident der ÖGSMP (2022). Außerdem ist er wiss. Beirat der sportärztezeitung.
© UWK/Daniel Novotny
Die trinationale (Deutschland, Österreich, Schweiz) Gesellschaft für Orthopädisch-Traumatologische Sportmedizin (GOTS) ist der größte europäische Zusammenschluss von Sportorthopäden und Sporttraumatologen. Sie ist erster Ansprechpartner in der Versorgung von Sportverletzungen und Garant für Qualität in der sporttraumatologischen Versorgung. Ihr Ziel ist es, das Verständnis von sportlicher Belastung und Verletzungen zu verbessern, um die muskuloskelettale Funktion und Lebensqualität zu erhalten. Dafür fördert die GOTS die Aus-und Weiterbildung, die Forschung sowie den internationalen Austausch unter sportorthopädisch und sporttraumatologisch tätigen Medizinern und Berufsgruppen angrenzender Fachgebiete.