Spinal pain is a very common reason for the limited ability to pursue sport for athletes in almost all types of sport. Therefore, concerning oneself with the vertebral column is very important in sports medicine for the locomotor apparatus. The use of PRP (platelet-rich plasma) as a therapeutic procedure for disorders of the locomotor apparatus is already widely established. PRP is finding increased acceptance as a promising minimally-invasive alternative form of treatment for the vertebral column.
In exactly the same way as throughout the entire locomotor apparatus, chronic inflammatory and degenerative changes play a decisive role as the cause of the relevant symptoms here, too. The now extensive relevant literature contains a high level of evidence in favour of treating osteoarthritis (e.g. osteoarthritis of the knee), [1, 2] tendinosis (e.g. tennis elbow, jumper’s knee, achillodynia) and ligamentosis [3 – 6]. Essentially similar structures and the corresponding pathological changes also play a decisive role in disorders of the spine. One of the main causes of spinal pain is segment degeneration in the lower lumbar spine where the high loads, often repetitive, imposed by sport activate inflammatory processes which lower the threshold of the nociceptive pain generators. Activated osteoarthritis of the lumbar and cervical facet joints, activated costotransverse joints, the SI syndrome and painful iliolumbar ligamentous apparatus are among the main indications for the use of PRP [7]. PRP is also successfully applied for discogenic and peri-/neural symptoms. The therapeutic benefit of PRP is based above all on its high proportion of growth factors released by the platelets. These have both anti-inflammatory and regenerative-proliferative effects on the affected tissues, and stimulate and accelerate healing processes [13, 21]. In my practice I use the low-leukocyte PRP from Arthrex, ACP (Autologous Conditioned Plasma), also because of its simple handling and the sterile double syringe system. A correct and exact diagnosis is essential for the efficacy of PRP treatment. Finding this can be a challenge, especially in the vertebral column. The pain focus and the pathological changes shown in the scans are correlated with the manual medicine examination. The affected anatomical structures of the spinal components (vertebral bodies, facet joints, nerve roots, intervertebral discs) and the related soft tissues (muscles, fasciae, ligaments) should all be identified as exactly as possible. The diagnosis can be verified by a therapeutic test infiltration. The pain relief the patient experiences contributes towards good patient compliance.
Treatment of the facet joints
Activated facet joint osteoarthritis is the most common indication for the use of PRP in the spine. This can be imaged well using a linear transponder (e.g. 9 to 12 MHz) in slender, athletic patients. A convex probe is used for deeper imaging (Fig. 1).
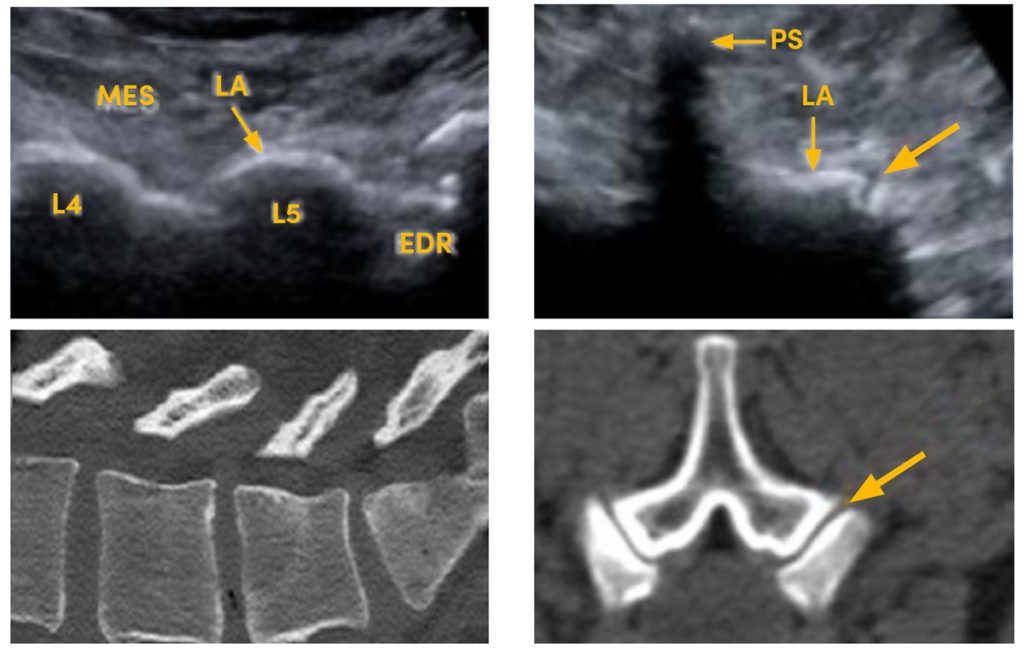
Fig. 1 It is essential to identify the affected structure in the spine and to apply the platelet-rich plasma exactly to the spot. Besides the established procedure using radiological guidance (computed tomography or fluoroscopy) ultrasonography has increasingly proved effective for guided injections: left, sagittal visualisation of the facet joints L3 to S1 and the indicated direction for infiltration on ultrasonography (top) and the computed tomography correlation (bottom). Right, analogously in transverse section L3 to S1 (PS = spinous process, LA = lamina, MES = erector spinae muscle, EDR = epidural space) EDR = Epiduralraum)
The cervical facet joints are very readily accessible to ultrasound. Joint effusion, synovial swelling and osteoarthritic changes are recognised during the diagnostic scan, correlated with the pain, and treated specifically (Fig. 2).
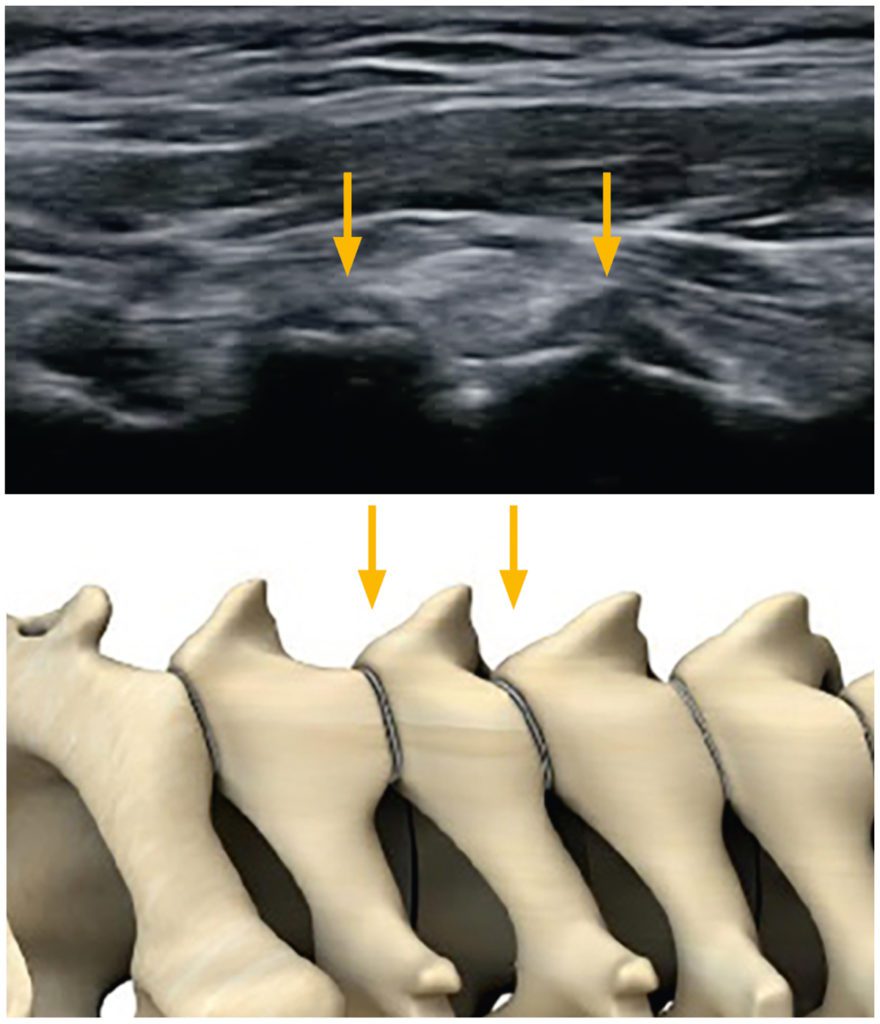
Fig. 2 Example of cervical facets; joint space C3/4 and C4/5 –>
In the case of pronounced inflammatory changes, corticosteroid injections are often successfully used to “calm down” joints, especially painfully activated facet joints. However, steroids do not prompt a healing process and moreover, they also cause chondral degeneration which limits their use [9]. Numerous studies confirm the better long-term effects of PRP treatment in direct comparison with steroids [2]. There is also corresponding evidence of this in the spine [10, 11]. In a comparison of the intra-articular use of corticosteroids with PRP in patients with facet syndrome, Wu et al. showed that pain perception in patients treated with steroids initially regressed but then increased again continually, while the pain perception decreased continually and significantly over the entire 6-month observation period in patients given treatment with PRP [12]. Similar superiority regarding the long-term effect of PRP treatment was also observed in the study by Braun et al. [13]. In my many years’ personal experience, the use of PRP on facet joints (intra- and periarticular) has very low rates of complications and side effects. PRP often has a longer-lasting effect on the patients treated. Nevertheless, the patient should be informed about possible initial exacerbation of the pain and delayed improvement (contrary to local anaesthetic / steroid injections). Alternatively, in view of advanced degenerative changes, for example in severe hypertrophic osteoarthritis of the facet joints, ablation of the “medial branches” (facet rhizotomy) must be considered for a good long-term outcome [14].
Case report cervical facets
At his first consultation at the end of 2016, the 56-year-old intensive amateur athlete (racing bike and kayak) complained of high right cervical pain radiating into the shoulder and arm. The MRI confirmed the diagnosis of severely activated osteoarthritis of the right C 2/3 facet joint (Fig. 3). The patient decided for treatment with ACP (low-leukocyte PRP). A series of five treatments at weekly intervals was able to reduce the symptoms almost completely. To date he has had one relapse which was again treated successfully with a series of three injections.
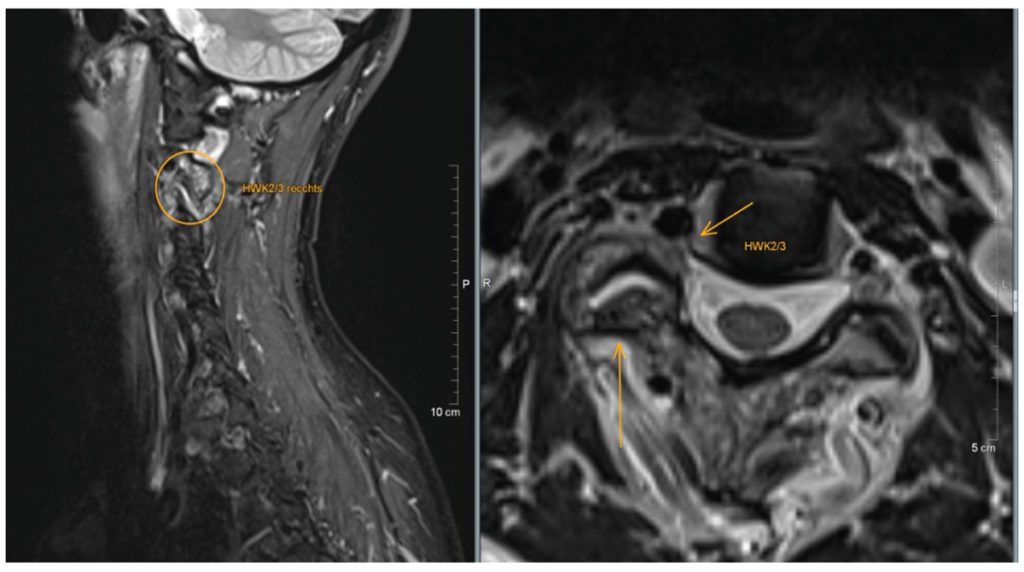
Fig. 3 MRI: isolated unilateral hypertrophic osteoarthritis of the right C 2/3 facet joint with activation demonstrated by bone marrow oedema, articular effusion and extensive periarticular soft-tissue oedema: higher-grade right foraminal stenosis as a consequence of this.
Treatment of the sacroiliac joints
Daily practice routine also shows good results in the use of ACP for disorders of the sacroiliac joint. The lower pole of the SI joint space is readily shown on ultrasound and easily infiltrated (Fig. 4). The literature reports better results of using PRP for the SI joints than with intra-articular methylprednisolone injections. In their randomised prospective study, Singla et al. enrolled a total of 40 patients and observed a long-lasting improvement in PRP patients as shown by established scores such as VAS and MODQ [7]. In my own experience, rheumatological inflammatory disorders of the sacroiliac joints respond well to PRP treatment.
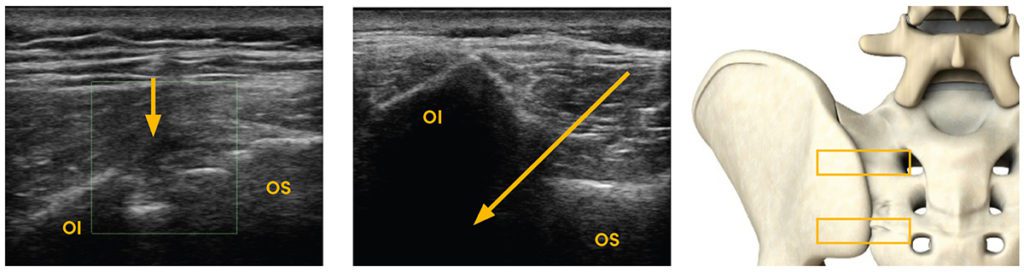
Fig. 4 Sacroiliac joint in a transverse sonogram: joint space and indicated direction for infiltration –> at the lower pole. Image 1 left and central, image 2 right (position of the probe indicated by the boxes); OI = ilium, OS = sacrum
Case Report SI
The 48-year-old recreational athlete came to my practice in 2015 with intensive predominantly buttock pain. In view of his typical medical history with alternating nocturnal pain at rest without any precipitating factors, morning stiffness and evidence of florid bilateral sacroiliitis with oedematous changes of the anterior vertebral bodies (shiny corners) in the MRI, the diagnosis of ankylosing axial spondylitis (Bechterew’s disease) was made. He was initially given steroid infiltrations into the sacroiliac joints, and later the TNF-α antagonist Humira (adalimumab) was established as a systemic treatment. All the steroid injections into the SI joints showed a very good effect, but as the pain returned after 5 – 6 months each time, the injections had to be repeated. In 2017 we switched to treating the SI joints on both sides with 5 mL ACP each as a series of three injections guided by ultrasonography. The patient definitely prefers this treatment to cortisone due to the “gentler and longer-lasting effect” – even if the PRP infiltration is more painful and does not offer instant relief. The injection series have been successfully repeated every 9 to 12 months to the present day. In some cases activated osteoarthritis of the facet joints of L4 to S1 and accompanying enthesiopathies (e.g. lateral epicondylitis) have to be treated as well.
Pelvic Ligamentosis
The chronically overloaded iliolumbar ligamentous apparatus responds very well to PRP in a multimodal treatment programme. Ligamentous instability and overload are an important cause of pain in the sacral region. This affects the iliolumbar, sacroiliac, sacrospinous and sacrotuberous ligaments [15, 16]. Hackett proposed the theory of musculoskeletal pain being caused by ligamentous laxity secondary to enthesiopathies [17]. Various factors such as muscular imbalance, weak posture, overloading and instability can trigger chronic inflammatory reactions in the periarticular structures as well as in the ligamentous apparatus [18]. Corticosteroid injections provide effective therapy for inflammation and local pain associated with ligamentous disorders. However, they also impair tissue regeneration. In ligaments and tendons with chronic inflammatory degenerative changes they inhibit fibroblast activation and, in turn, collagen synthesis required for healing [19]. Collagen necrosis at the injection site is possible [20]. Therefore, multiple use of local steroids is not advisable. In contrast, alternative PRP therapy can induce regenerative processes and can be recommended as an injectable for modern prolotherapy. Precisely in connection with this, PRP therapy must be embedded in a multimodal treatment programme to counteract the cause of overload. An exercise programme to improve posture and to recondition and improve muscular torso stability should be established. Loading exercises lead to efficient and successful remodelling in the affected and treated structure.
Summary
Due to the very promising results, both in daily use and in the literature, the focus is turning increasingly to treatment with platelet-rich plasma for spinal symptoms. Activated osteoarthritis of the lumbar and cervical facet joints, sacroiliac syndrome and the painful iliolumbar ligamentous apparatus are among the main indications for the use of PRP. Activation of regenerative processes leads to long-lasting pain relief and improved function. According to the literature, the long-term outcomes are better than those with corticosteroid injections. Successful treatment is based on a correct diagnosis and targeted treatment of the affected structure. Ultrasound enables simple, quick and specific administration to many sites in the spine. In my own practice, ACP from the Arthrex company has proved its worth due to the low risk use that is practically free of side effects. To this extent, PRP treatment should be embedded in a multimodal treatment programme wherever possible.
References
[1] P. A. Smith, “Intra-articular Autologous Conditioned Plasma Injections Provide Safe and Efficacious Treatment for Knee Osteoarthritis,” Am. J. Sports Med., vol. 44, no. 4, pp. 884–891, 2016, doi: 10.1177/0363546515624678.
[2] Y. Huang, X. Liu, X. Xu, and J. Liu, “Intra-articular injections of platelet-rich plasma, hyaluronic acid or corticosteroids for knee osteoarthritis: A prospective randomized controlled study,” Orthopade, vol. 48, no. 3, pp. 239–247, 2019, doi: 10.1007/s00132-018-03659-5.
[3] T. E. Foster, B. L. Puskas, B. R. Mandelbaum, M. B. Gerhardt, and S. A. Rodeo, “Platelet-rich plasma: From basic science to clinical applications,” American Journal of Sports Medicine. 2009, doi: 10.1177/0363546509349921.
[4] E. Kon et al., “Platelet-rich plasma : intra-articular knee injections produced favorable results on degenerative cartilage lesions,” pp. 472–479, 2010, doi: 10.1007/s00167-009-0940-8.
[5] A. Mishra and T. Pavelko, “Treatment of chronic elbow tendinosis with buffered platelet-rich plasma,” Am. J. Sports Med., 2006, doi: 10.1177/0363546506288850.
[6] M. De Mos et al., “Can platelet-rich plasma enhance tendon repair? A cell culture study,” Am. J. Sports Med., 2008, doi: 10.1177/0363546508314430.
[7] V. Singla, Y. K. Batra, N. Bharti, V. G. Goni, and N. Marwaha, “Steroid vs. Platelet-Rich Plasma in Ultrasound-Guided Sacroiliac Joint Injection for Chronic Low Back Pain,” Pain Pract., vol. 17, no. 6, pp. 782–791, Jul. 2017, doi: https://doi.org/10.1111/papr.12526.
[8] J. Yamada, K. Akeda, N. Takegami, K. Nakase, T. Sano, and A. Sudo, “Anti-inflammatory properties of platelet rich plasma-releasate on human intervertebral disc cells,” J. Orthop. Res. Conf., 2017.
[9] T. E. McAlindon et al., “Effect of intra-articular triamcinolone vs saline on knee cartilage volume and pain in patients with knee osteoarthritis a randomized clinical trial,” JAMA – J. Am. Med. Assoc., 2017, doi: 10.1001/jama.2017.5283.
[10] T. J. Wu, C. Y. Hung, C. W. Lee, S. Lam, T. B. Clark, and K. V. Chang, “Ultrasound-guided lumbar Intradiscal injection for Discogenic pain: Technical innovation and presentation of two cases,” J. Pain Res., vol. 13, pp. 1103–1107, May 2020, doi: 10.2147/JPR.S253047.
[11] Tuakli-Wosornu et al., “Lumbar Intradiskal Platelet-Rich Plasma (PRP) Injections: a Prospective, Double-Blind, Randomized Controlled Study,” PM r, 2016.
[12] J. Wu et al., “A Prospective Study Comparing Platelet-Rich Plasma and Local Anesthetic (LA)/Corticosteroid in Intra-Articular Injection for the Treatment of Lumbar Facet Joint Syndrome,” Pain Pract., vol. 17, no. 7, pp. 914–924, 2017, doi: 10.1111/papr.12544.
[13] H. J. Braun, H. J. Kim, C. R. Chu, and J. L. Dragoo, “The effect of platelet-rich plasma formulations and blood products on human synoviocytes: Implications for intra-articular injury and therapy,” Am. J. Sports Med., 2014, doi: 10.1177/0363546514525593.
[14] P. Dreyfuss, B. Halbrook, K. Pauza, A. Joshi, J. McLarty, and N. Bogduk, “Efficacy and validity of radiofrequency neurotomy for chronic lumbar zygapophysial joint pain,” Spine (Phila. Pa. 1976)., 2000, doi: 10.1097/00007632-200005150-00012.
[15] A. Pool-Goudzwaard, G. H. Van Dijke, P. Mulder, C. Spoor, C. Snijders, and R. Stoeckart, “The iliolumbar ligament: Its influence on stability of the sacroiliac joint,” Clin. Biomech., 2003, doi: 10.1016/S0268-0033(02)00179-1.
[16] T. Aihara, K. Takahashi, A. Ogasawara, E. Itadera, Y. Ono, and H. Moriya, “Intervertebral disc degeneration associated with lumbosacral transitional vertebrae. A clinical and anatomical study,” J. Bone Jt. Surg. – Ser. B, 2005, doi: 10.1302/0301-620X.87B5.15727.
[17] H. G., “Low Back Pain,” Br. J. Phys. Med., vol. 19, pp. 25–35, 1956.
[18] P. D. Palesy, “Tendon and ligament insertions – A possible source of musculoskeletal pain,” Cranio, 1997, doi: 10.1080/08869634.1997.11746012.
[19] H. Oxlund, “the influence of a local injection of cortisol on the mechanical properties of tendons and ligaments and the indirect effect on skin,” Acta Orthop., 1980, doi: 10.3109/17453678008990791.
[20] U. Fredberg, “Local corticosteroid injection in sport: Review of literature and guidelines for treatment,” Scandinavian Journal of Medicine and Science in Sports. 1997, doi: 10.1111/j.1600-0838.1997.tb00129.x.
[21] A. D. Mazzocca et al., “The positive effects of different platelet-rich plasma methods on human muscle, bone, and tendon cells,” Am. J. Sports Med., vol. 40, no. 8, pp. 1742–1749, 2012, doi: 10.1177/0363546512452713.
Autoren
leitet die Praxis Orthopädie am Rhy in Rheinfelden und ist spezialisiert auf konservative und interventionelle Behandlungsmethoden am ganzen Bewegungsapparat. Er hat die Zusatzbezeichnungen Sportmedizin, Ultraschall und interventionelle Schmerztherapie sowie manuelle Medizin und Notfallmedizin. Dr. Dau hat in der Schweiz diverse Sportteams betreut und langjährige Erfahrung in der Behandlung von Profi- und Spitzensportlern.